-
[ June 11, 2012]
Uncovering Molecular Mechanisms of Electrowetting and Saturation with Simulations
-
Peking University professors have teamed up with scientists from Johns Hopkins University and Tsinghua University in a research to uncover molecular mechanisms of electrowetting and saturation with simulations. Their work was published in Physical Review Letters on May 21, 2012.
In electrowetting, an applied voltage changes the macroscopic contact angle of a liquid drop on a solid. While electrowetting has proved useful in manipulating small drops, applications are limited because the contact angle always saturates as the applied voltage increases.
The cause of saturation and the physics underlying electrowetting have been the subject of active debate, and previous theories have been limited to continuum models that become inadequate as drops shrink to micrometer or nanometer scales.
Shiyi Chen, dean of the Peking University College of Engineering and a professor in the State Key Laboratory for Turbulence and Complex Systems (SKLTCS), and his cooperators used molecular simulations to explore the atomistic underpinnings of electrowetting and the limits of continuum theory.
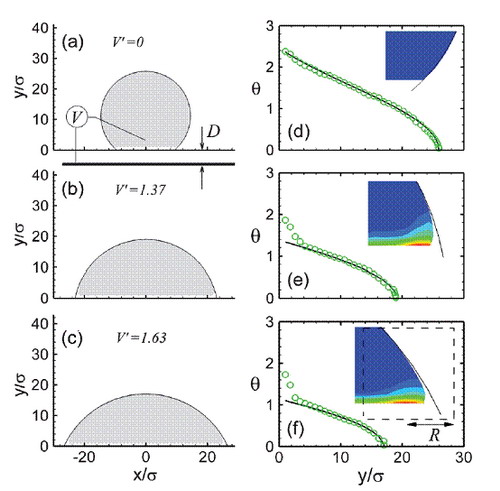
The figure shows how the drop shape changes with voltage and the distribution of charge
The results show that Nanoscale drops exhibit the same behavior seen in macroscopic experiments. Initially, the decrease in contact angle with increasing applied voltage follows continuum theory, but the contact angle saturates at large applied voltage.The research team also found that saturation occurs when charged molecules are pulled from the drop and is controlled by local electric field strengths and molecular binding. An alternative expression for contact angle based on local force balance remains valid even after saturation and explains why the contact angle approaches equilibrium state within a few nanometers of the solid.
Paper cited:
Uncovering Molecular Mechanisms of Electrowetting and Saturation with Simulations, Phys. Rev. Lett. 108, 216101(2012)
