-
[ October 09, 2012]
Break-through in controlled synthesis of Fe5C2 magnetic nanoparticles and their application in new energy conversion
-
Recently, Material Scientist Yanglong Hou, a professor in the Department of Materials Science and Engineering College of Engineering (COE), achieved a breakthrough in controlled synthesis of Fe5C2 magnetic nanoparticles and their application in new energy conversion, by collaborating with Professor Ding Ma from the College of Chemistry and Molecular Engineering (CCME), Peking University. The results were published online in an article titled “Fe5C2 Nanoparticles: A Facile Bromide-induced Synthesis and as an Active Phase for Fischer - Tropsch Synthesis (FTS)” in Journal of American Chemical Society ( http://pubs.acs.org/doi/abs/10.1021/ja305048p ).
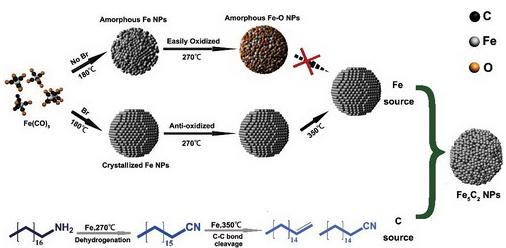
Synthetic mechanism of Fe5C2 NPs
For centuries, iron carbides have gained intense interest both in fundamental science and in applied engineering. Primarily, iron carbides consist of carbon atoms occupying the interstices between close-packed iron atoms. The presence of carbon atoms provides iron carbides with excellent mechanical strength and chemical inertness. In fact, the discovery of iron carbides in ancient Wootz steels in India indicated that iron carbides had been widely used as reinforcement materials 2000 years ago. In contemporary studies, several unique properties have been found of iron carbide nanostructures including superparamagnetism, catalytic activity and so on, which provide Fe5C2 nanostructures with great potential in application of biomedicine and catalysis.
Fischer?Tropsch synthesis (FTS) can be expressed as nCO + (2n + 1)H2 → CnH2n+2 + nH2O. Since the ?rst report in 1923, FTS has been considered to be a practical approach for producing liquid fuels from fossil resources such as natural gas and coal as well as biomass-derived biogas. And a majority of reports have suggested that iron carbides are possibly the active phases of iron-based catalysts. However, iron carbide nanostructures have been rarely investigated, primarily because there are still big challenges in the synthetic strategy of iron carbide nanostructures.
Recently, Hou and co-workers reported a facile chemie douce route for the synthesis of Fe5C2 NPs that involves the reaction of iron carbonyl, Fe(CO)5, with octadecylamine in the presence of bromide under mild temperatures (up to 623 K). The size of the iron carbide NPs can be tuned by tailoring the concentration of Fe(CO)5. The as-synthesized Fe5C2 NPs were used as an FTS catalyst, and the results demonstrated that Fe5C2 possesses higher activity and selectivity than a conventional reduced-hematite catalyst. More importantly, the induction period observed with conventional iron oxide catalysts was not observed with this catalyst, which clearly indicates that Fe5C2 is an active phase for FTS.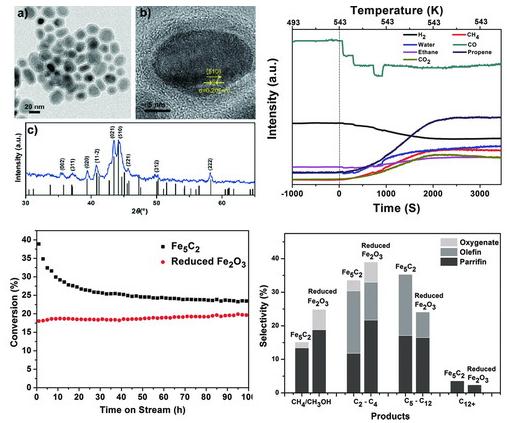
The structural and catalytic characterization of Fe5C2 NPs
The first author of this article is Ce Yang, a 1st year Ph.D. candidate in Hou’s group in COE. The second author is Huabo Zhao, a 3rd year Ph.D. candidate in Ma’s group in CCME. This project is supported by the National Science Foundation of China and the National Basic Research Program of China.
