-
[ July 25, 2014]
Dr. Antony K. Chen and collaborators make new progress in the research of microRNAs
-
Recently, Dr. Antony K Chen, an assistant professor of the Department of Biomedical Engineering, College of Engineering, together with Dr. Jennifer Lippincott-Schwartz of the National Institutes of Health and Dr. Eric Freed of the National Cancer Institute, discovered a novel mechanism by which microRNAs can inhibit HIV-1 virus production. The new mechanism involves interactions between microRNA and HIV-1 Gag protein’s RNA-binding (nucleocapsid) domain. The research was published in the Proceedings of the National Academy of Sciences (PNAS) (http://www.pnas.org/content/early/2014/06/16/1408037111.full.pdf+html).
The final step of HIV’s life cycle, the assembly of virus particles, begins at the plasma membrane within the host cell. Central to this process is the multimerization of Gag, the main structural protein that forms the shell of the HIV-1 virion. To trigger viral budding, Gag proteins must extensively multimerize at the plasma membrane, forming a tightly-packed lattice that remodels into a virus particle containing several thousand Gag molecules. Increasing evidence suggests that Gag multimerization is facilitated by long-stranded RNAs, including viral and cellular mRNAs, which serve as a scaffold for concentrating Gag proteins by binding nonspecifically to Gag’s nucleocapsid (NC) domain. The proposed role of nonspecific Gag–RNA interactions in facilitating Gag multimerization and viral budding prompted Chen et al., to investigate whether miRNAs, a class of small non-coding RNAs that are 18-22 in length, could prevent virus production by competing with viral RNA for Gag binding.
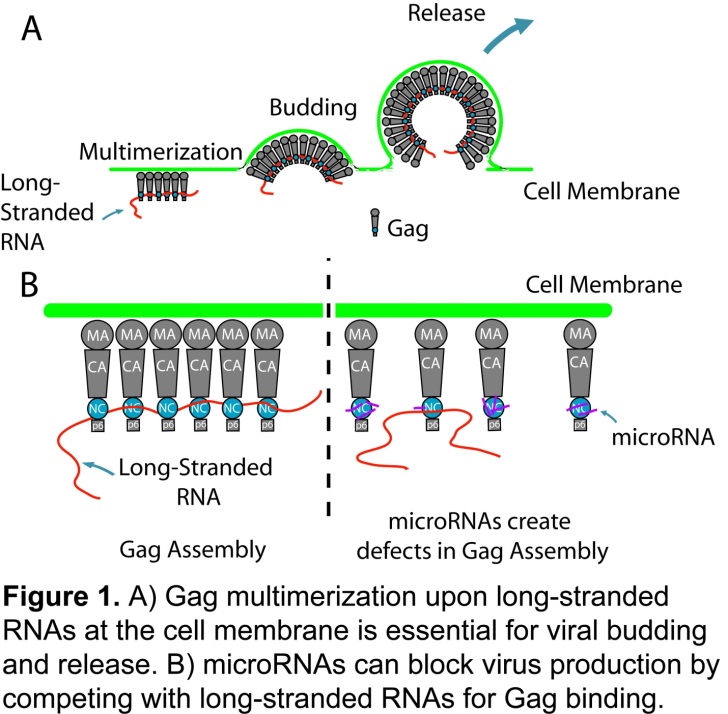
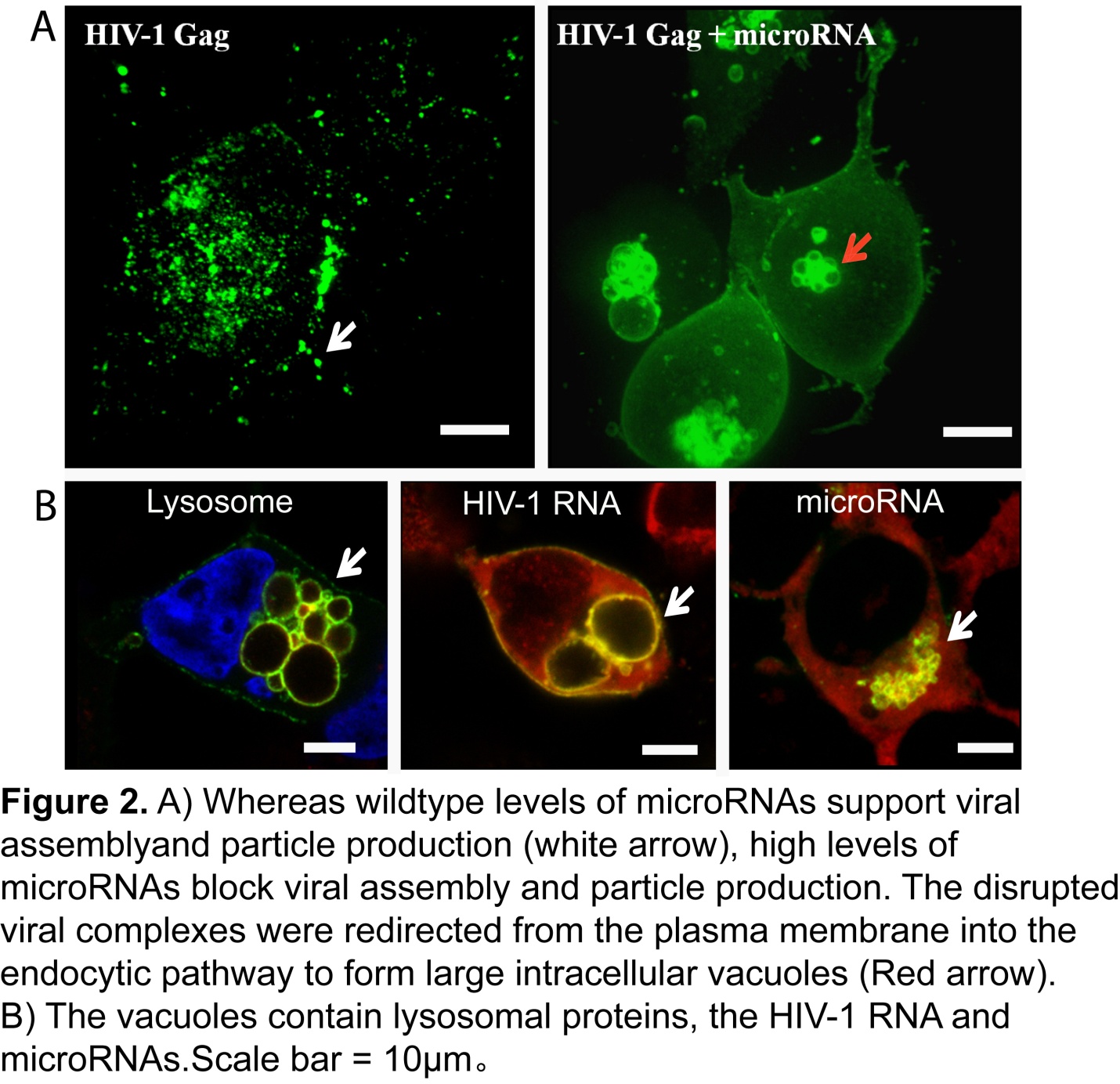
Using a myriad of live-cell imaging, biochemistry, RNA imaging and superresolution imaging techniques, Chen et al., demonstrated that Gag-microRNA interactions prevent Gag proteins from effectively multimerizing upon the HIV-1 viral RNA at the plasma membrane and lead to inhibition of viral particle production. Consequently, the disrupted viral complexes were redirected into the endocytic pathway and ultimately degraded in lysosomes. Overall, these findings have significant implications for understanding how cells modulate HIV-1 infection by miRNA expression and raise the possibility that miRNAs can function to disrupt RNA-mediated protein assembly processes in other cellular contexts.
Dr. Antony K. Chen is the first author of the paper. This project was supported in part by the National Natural Science Foundation of China.
