-
[ March 14, 2013]
Ying Luo makes progress in bio-material function studies
-
Recently, Dr. Ying Luo, associate professor in the Department of Biomedical Engineering, and her research team published two papers on Biomaterials about their studies of the application of dendrimer materials, after exploring nanocomposites’ capability of trans missing biological signal to cells.
Research work I
Dendrimers are a unique category of nanoscale materials that possess attractive structural features that include monodisperse molecular weight, tunable sizes and nanoscale starburst branches. By presenting biomolecular ligands on the surface in high density, ligand-decorated dendrimers are capable of binding to membrane receptors and cells with specificity and avidity.However, despite the various uses, fundamental investigations on ligand-dendrimer conjugates have mainly focused on their binding behavior with cells, whereas their potential bioactivity and applications in multicellular systems, especially in three-dimensional (3D) culture systems, remains untapped.
In their study, a typical adhesive peptide ligand – RGD – was modified to generation 4 polyamidoamine (PAMAM), and the bioactivity of suspended RGD-PAMAM conjugates was investigated on cells cultured as multicellular spheroids. Their results demonstrate that the RGD-PAMAM conjugates, after being incorporated into the 3D spheroids, were able to promote cellular proliferation and aggregation, and affect the mRNA expression of extracellular factors by NIH 3T3 cells. These bioactive functions were multivalency-dependent, as none of similar effects was observed for monovalent RGD ligand. Their study suggests that multivalent ligand-dendrimer conjugates may act as a unique type of artificial factors to mediate the cellular microenvironment in 3D culture, a property attributable to the spatial organization of the ligands and possible “cell-gluing” function of multivalent conjugates.
The results were published in paper “The effects of an RGD-PAMAM dendrimer conjugate in 3D spheroid culture on cell proliferation, expression and aggregation” (link). The experiments were jointly carried out by Ph.D. candidate Liyang Jiang and master student Bing Lv.
This new finding opens the door for further exploring multivalent ligand-dendrimer conjugates for applications in 3D cell culture and tissue engineering.
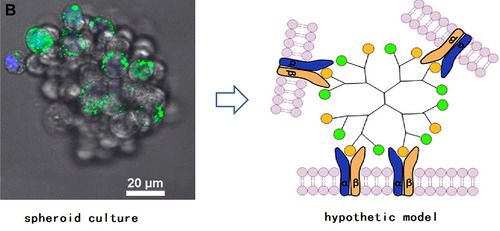
Incorporation of RGD-PAMAM conjugates in fibroblast spheroid cultureResearch work II
Cardiovascular disease (CVD) is the leading cause of death throughout the world and much pathology is associated with upregulation of inflammatory genes. Gene silencing using RNA interference is a powerful tool in regulating gene expression, but its application in CVDs has been prevented by the lack of efficient delivery systems.To further the studies, PKU Associate Professor Ying Luo joined hands with Dr. Mike Davis of the Wallace H. Coulter Department of Biomedical Engineering at Emory University and Georgia Institute of Technology. The team reports the development of tadpole dendrimeric materials for siRNA delivery in a rat ischemia-reperfusion (IR) model. Angiotensin II (Ang II) type 1 receptor (AT1R), the major receptor that mediates most adverse effects of Ang II, was chosen to be the silencing targeting. Among the three tadpole dendrimers synthesized, the oligo-arginine conjugated dendrimer loaded with siRNA demonstrated effective down-regulation in AT1R expression in cardiomyocytes in vitro. When the dendrimeric material was applied in vivo, the siRNA delivery prevented the increase in AT1R levels and significantly improved cardiac function recovery compared to saline injection or empty dendrimer treated groups after IR injury.
The results were published in paper “Functionalized dendrimer- based delivery of angiotensin type 1 receptor siRNA for preserving cardiac function following infarction” (link). PKU-GT-Emory Joint Ph.D. candidate Jie Liu who carried out the experimental research was the first author.
These experiments demonstrate a potential treatment for dysfunction caused by IR injury and may represent an alternative to AT1R blockade.
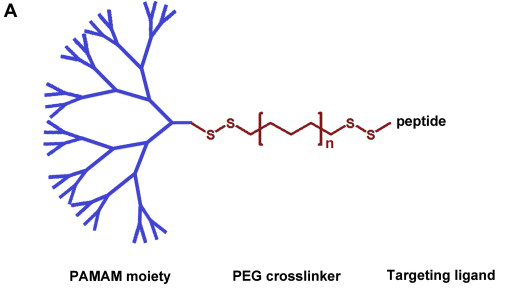
Tadpole design. The structure of the tadpole dendrimers
